Spring is Here! | CMI Health

The season we all love and hate.
Spring is here which means allergy season is on the rise. Allergies commonly arise during the spring season as a response to excessive pollen produced by outdoor plants. Allergy symptoms vary from person to person and can range from mild irritation to extreme breathing difficulties (Mayo Clinic, 2022). These symptoms arise due to an autoimmune response driven by increased IgE antibody production (antibodies responsible for combatting foreign substances). Though certain allergens can produce an immune response in different individuals, pollen is one of the most common culprits of reaction. Given that pollen is a small and excessively produced plant-derived particle, it can be easily inhaled, resulting in respiratory and ocular (eye) inflammation. During peak allergy season, dealing with the symptoms can be frustrating and, at times, overwhelming. Common spring allergies include:
- Sneezing
- Itchy nose, eyes, and mouth
- Runny, stuffy nose
- Swollen and red eyes
- Difficulties breathing
While there is no cure for allergies, daily medicinal treatment and lung management can help alleviate symptoms. For those who enjoy being outdoors often, or those previously diagnosed with chronic respiratory diseases, such as Asthma and COPD, monitoring daily lung function may be an ideal way to combat seasonal irritants.
Keep scrolling for information on devices designed to make managing your allergies simpler!

Dealing with chronic lung issues, such as Asthma and COPD, can be frustrating. Seasonal allergens can further exacerbate breathing complications, resulting in increased inflammation and irritation. Even so, monitoring daily vitals is an easy and efficient way to track lung function and maintain health awareness. By observing trends in respiratory health, users can identify key indicators of worsening symptoms before they become visible.
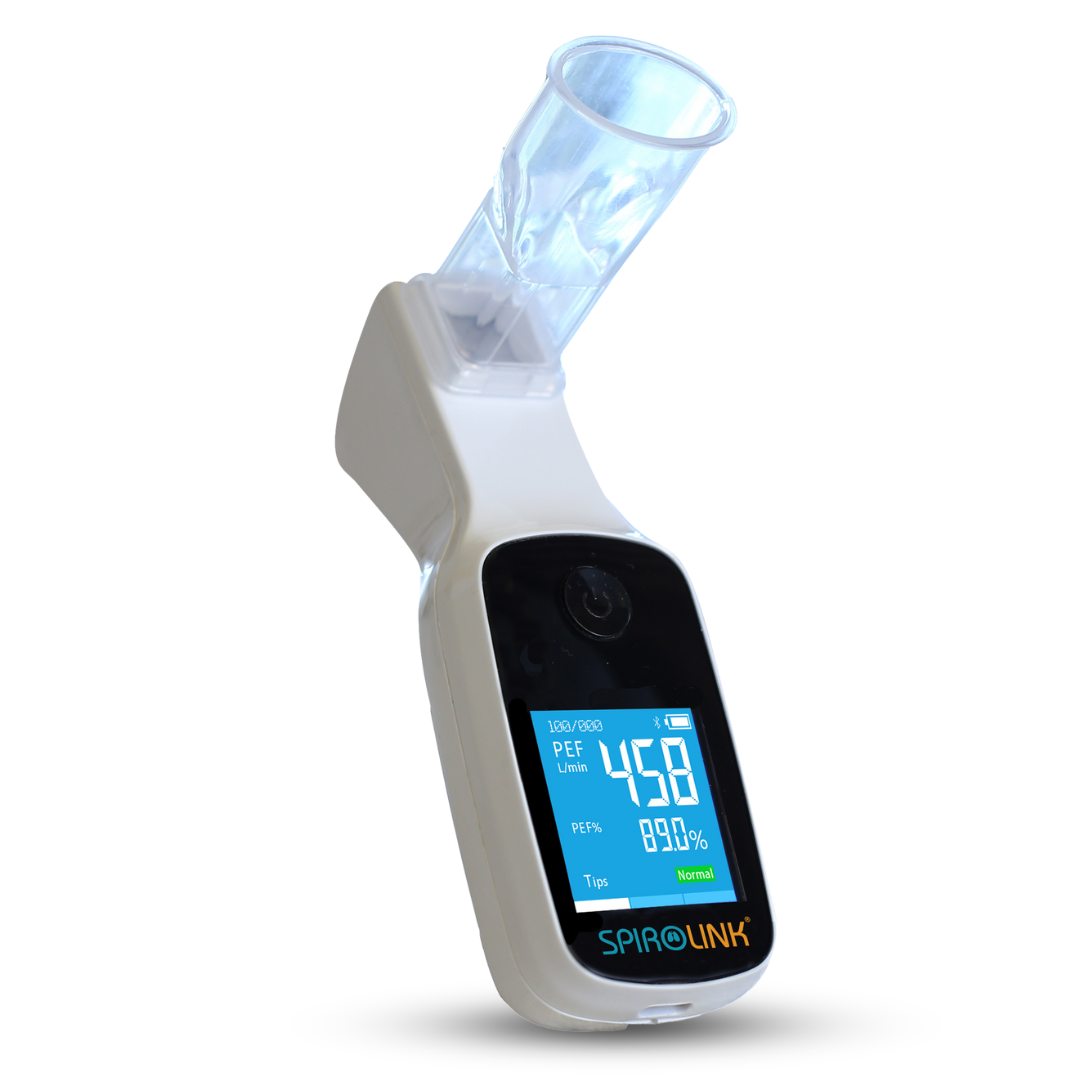
Through the use of SpiroLink®, our clinical-grade digital spirometer, users can take control of their at-home health monitoring. Spirometry is an important clinical tool used to measure and test an individual’s pulmonary function. Essentially, spirometry is a test used to determine how well a person's lungs are working. It does this by measuring how much, and how quickly an individual is able to exhale air from their lungs. SpiroLink's® Bluetooth capabilities allow a user to easily track and manage their spirometry data. It is especially well suited for daily measurement and management of lung health for a variety of users including those with asthma, COPD, musicians, athletes, smokers, and more.
By taking daily spirometry measurements, users can view and counter the effects of spring allergens on their overall lung function. Take control of your lung health this spring by investing in a one-of-a-kind spirometer you can rely on!

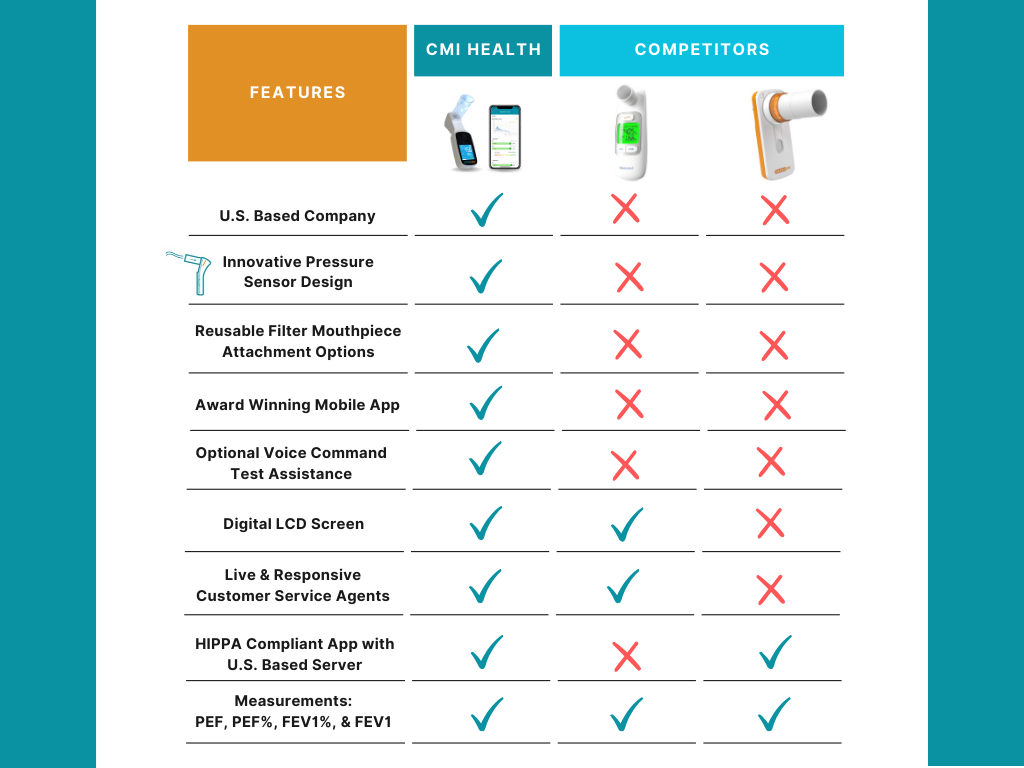
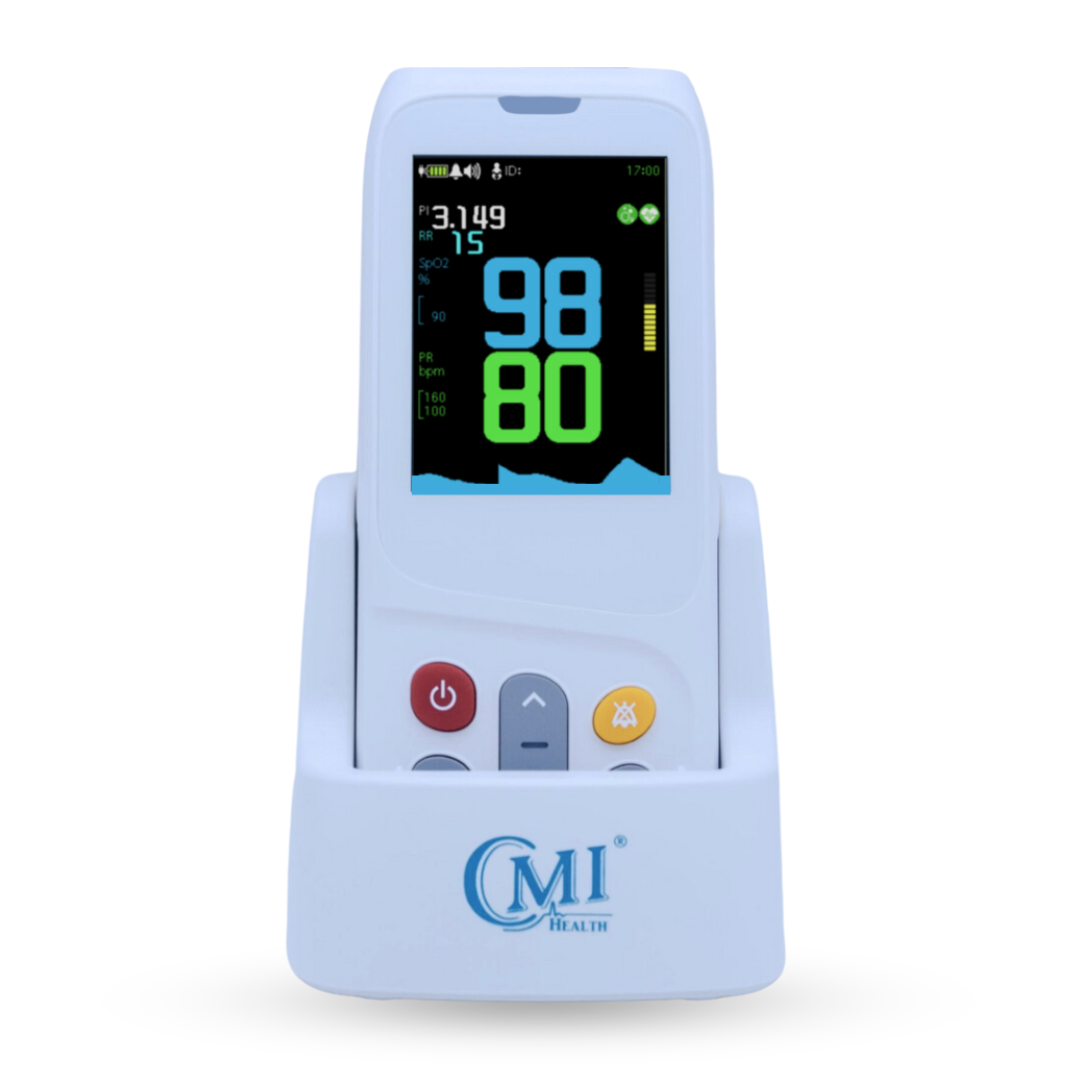
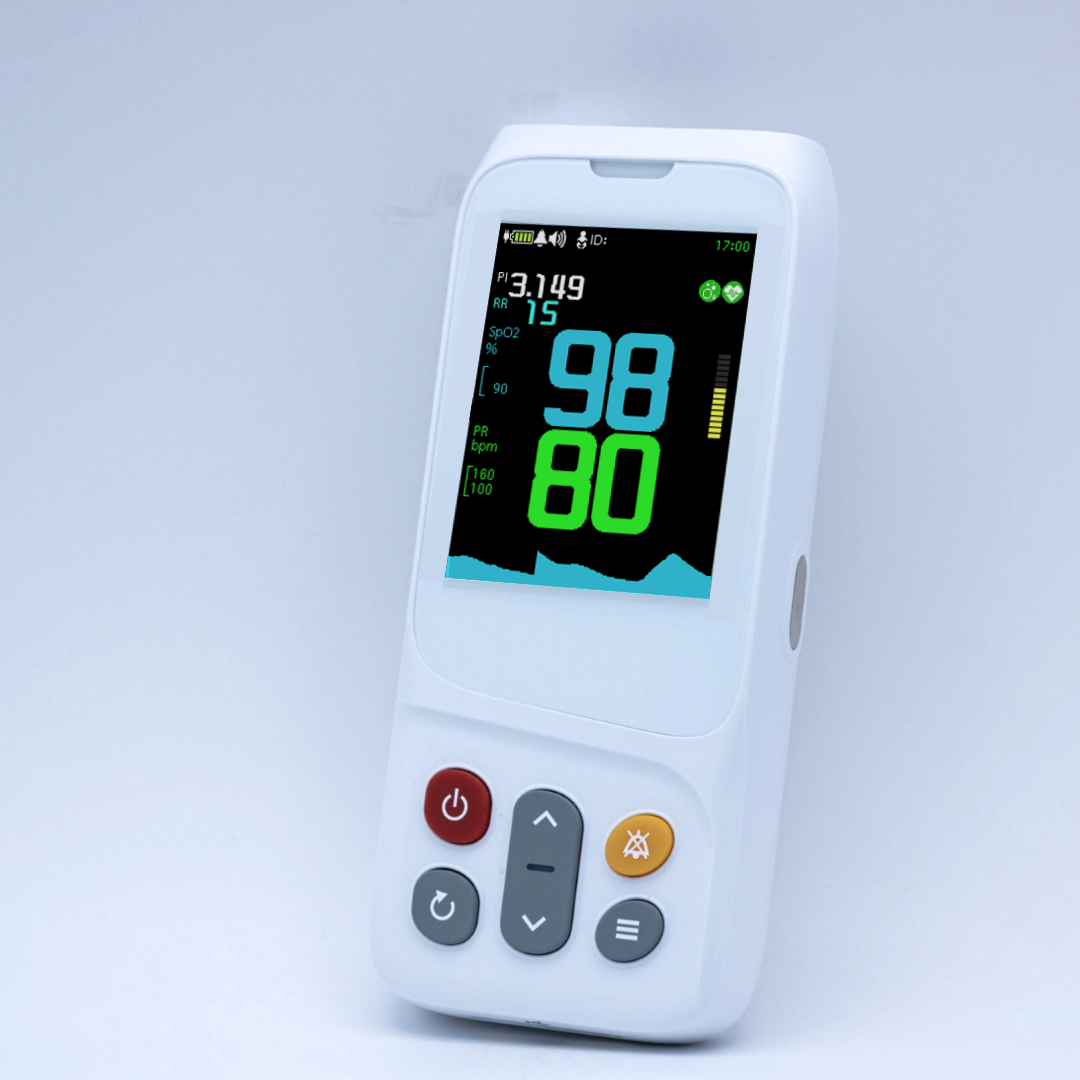
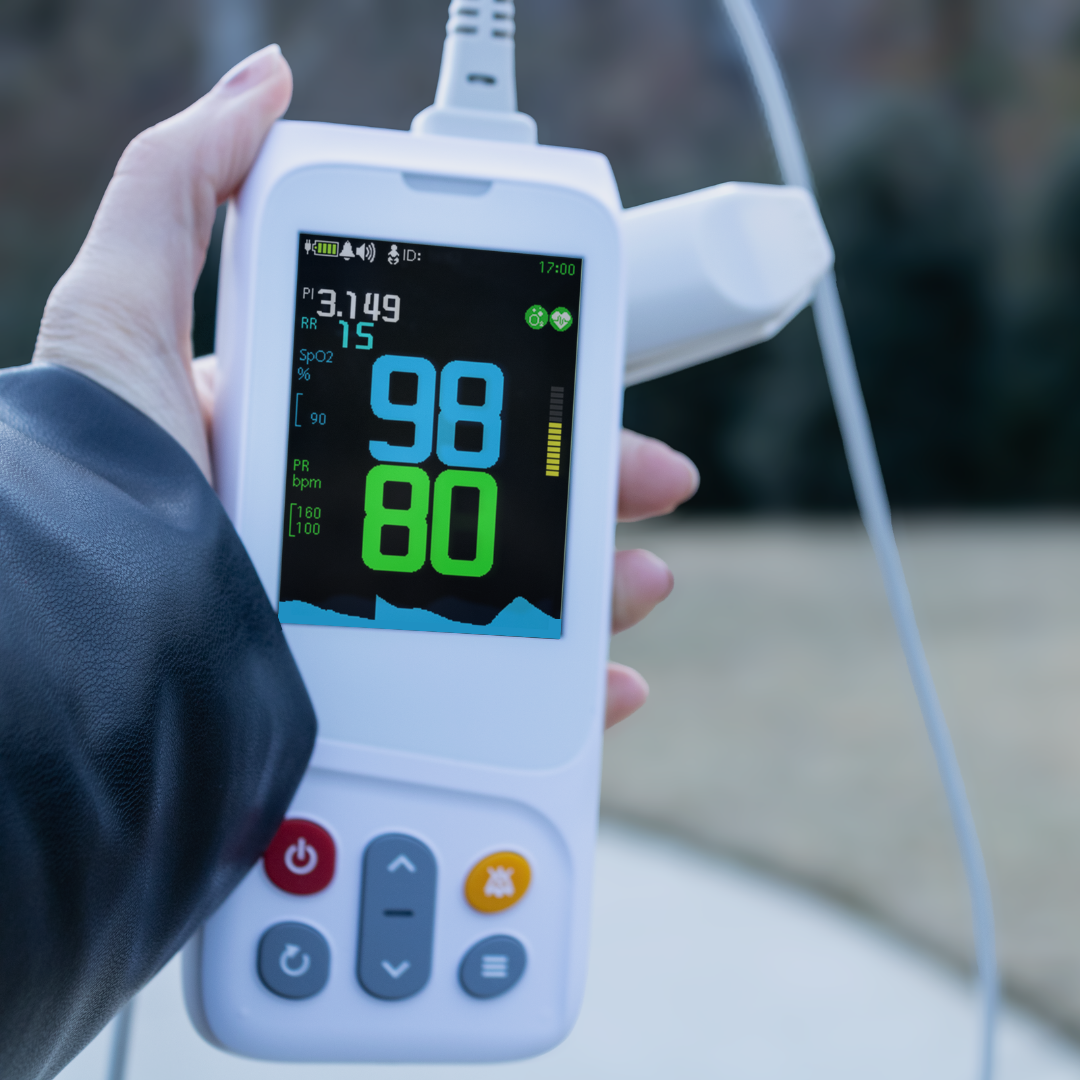
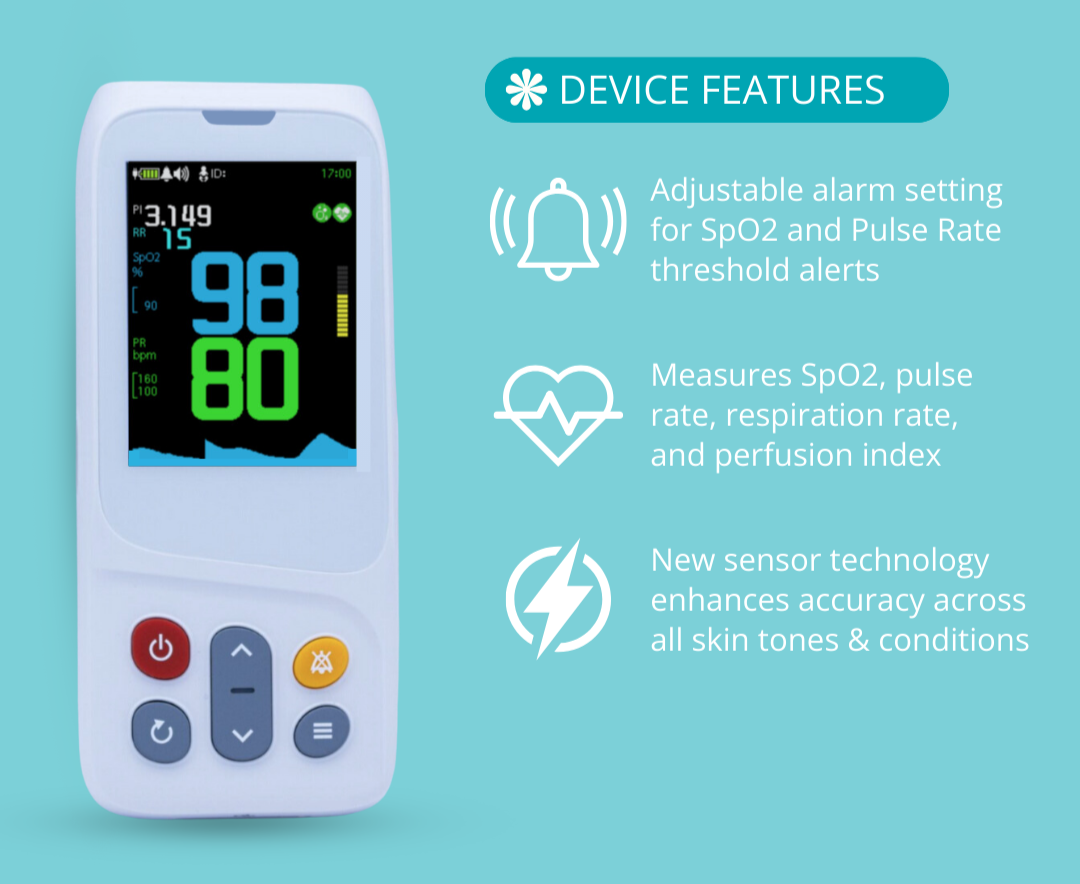
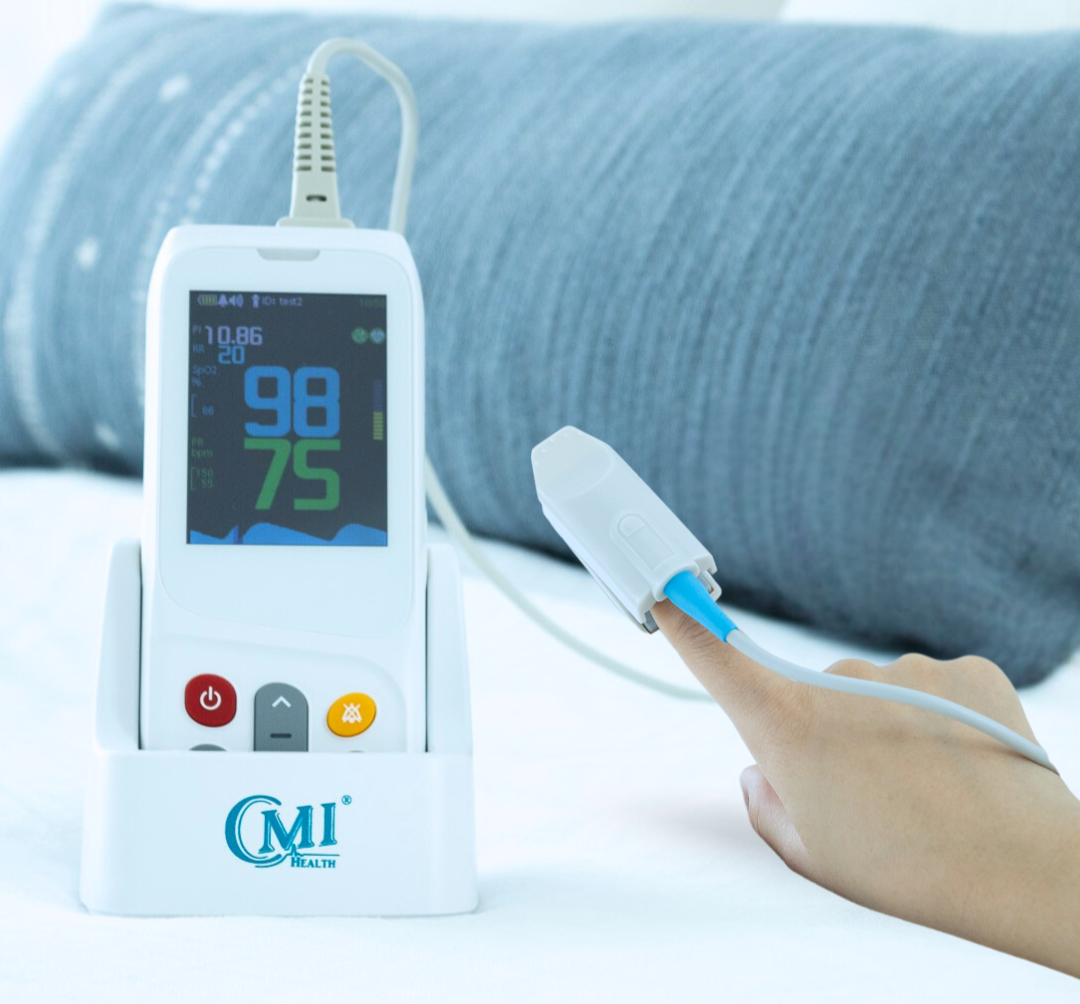
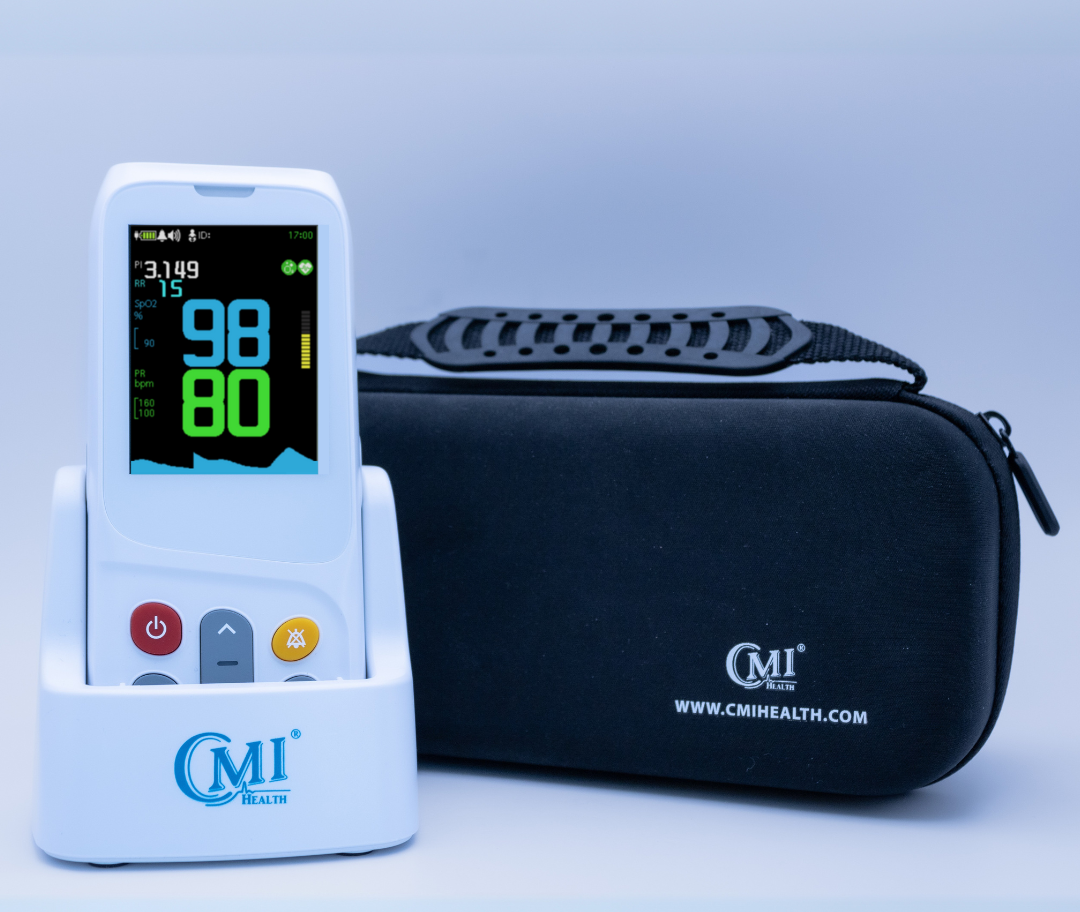
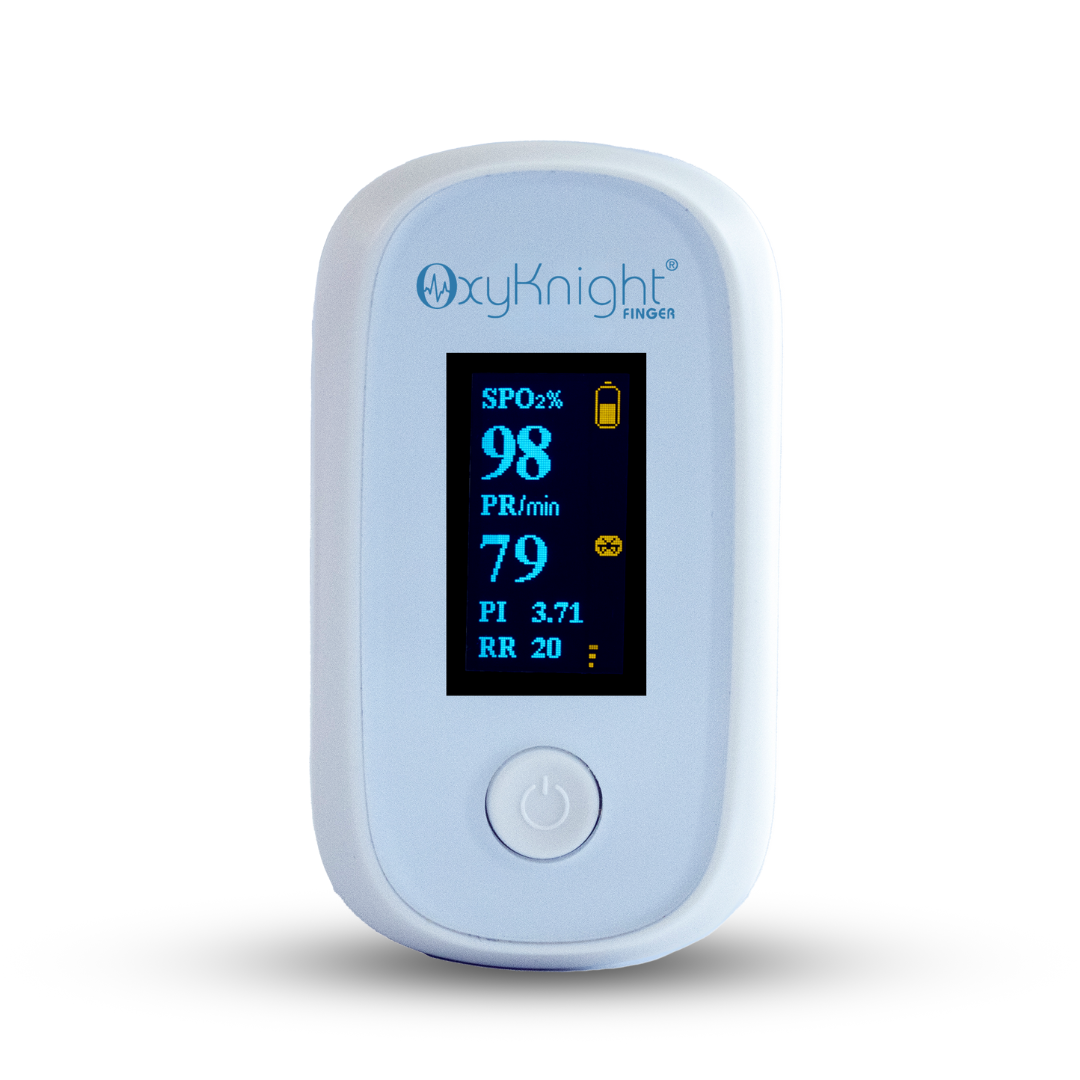
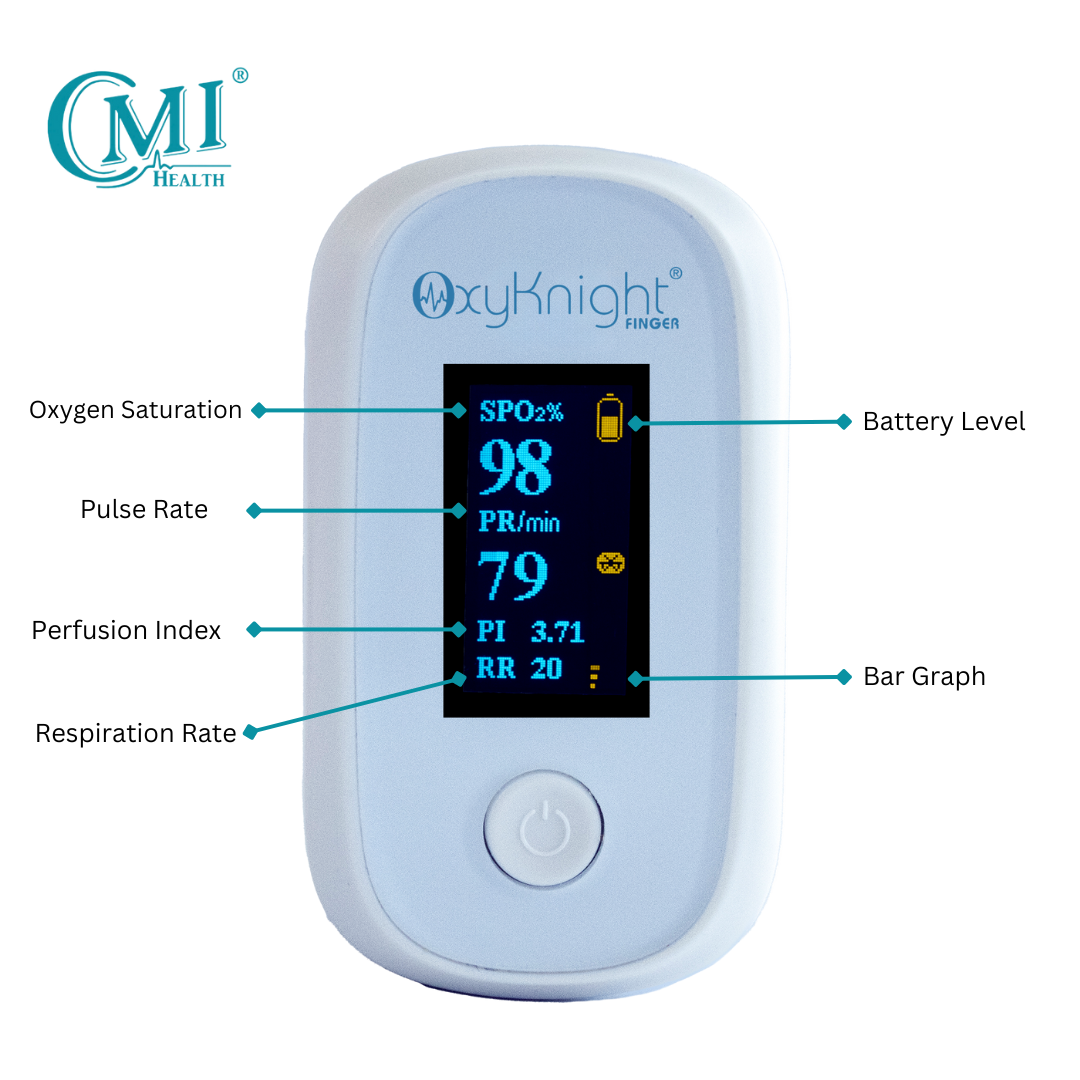
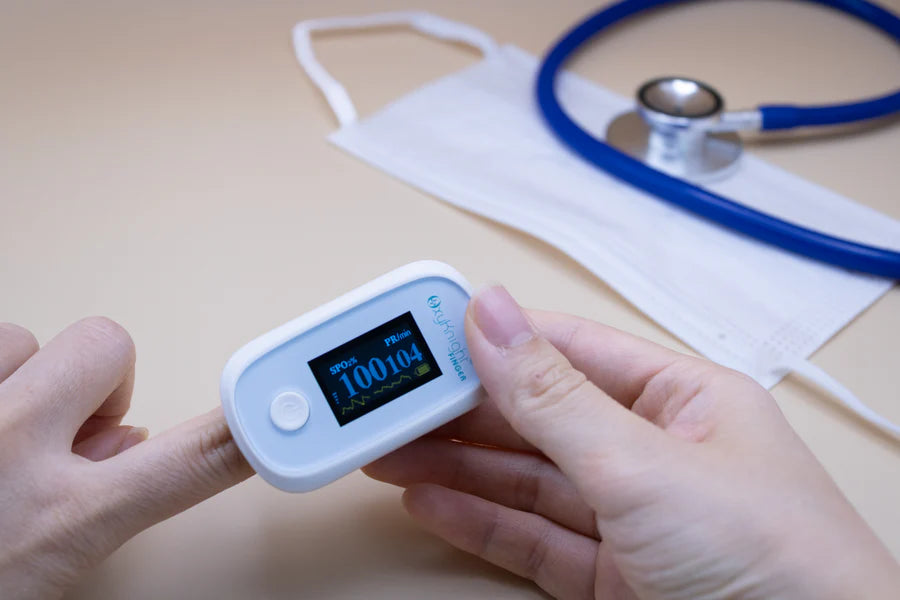
AsthmaGo™ is revolutionizing the way we approach at-home Asthma lung health management. By combining the use of 3 exceptional medical devices, users are able to get a clear and accurate view of their daily lung performance in one all-inclusive app. By tracking key lung performance data within the app, users can download and share their reports and respiratory trends with physicians remotely. Users are also able to take the Asthma Control Test (ACT) within the app, improving their ability to self-manage.
Users can log their daily symptoms, medication use, and more, allowing for enhanced self-management of personal diet, exercise, and lifestyle habits. Activation of low oxygen and high pulse rate alarms can alert the user to an oncoming or ongoing Asthma attack. Such warnings can result in faster response times and may help to prevent more serious complications. Check out the 3 devices that can be paired with AsthmaGo™ below!























Leave a comment