The Importance of Purchasing Quality Pulse Oximeters | CMI Health

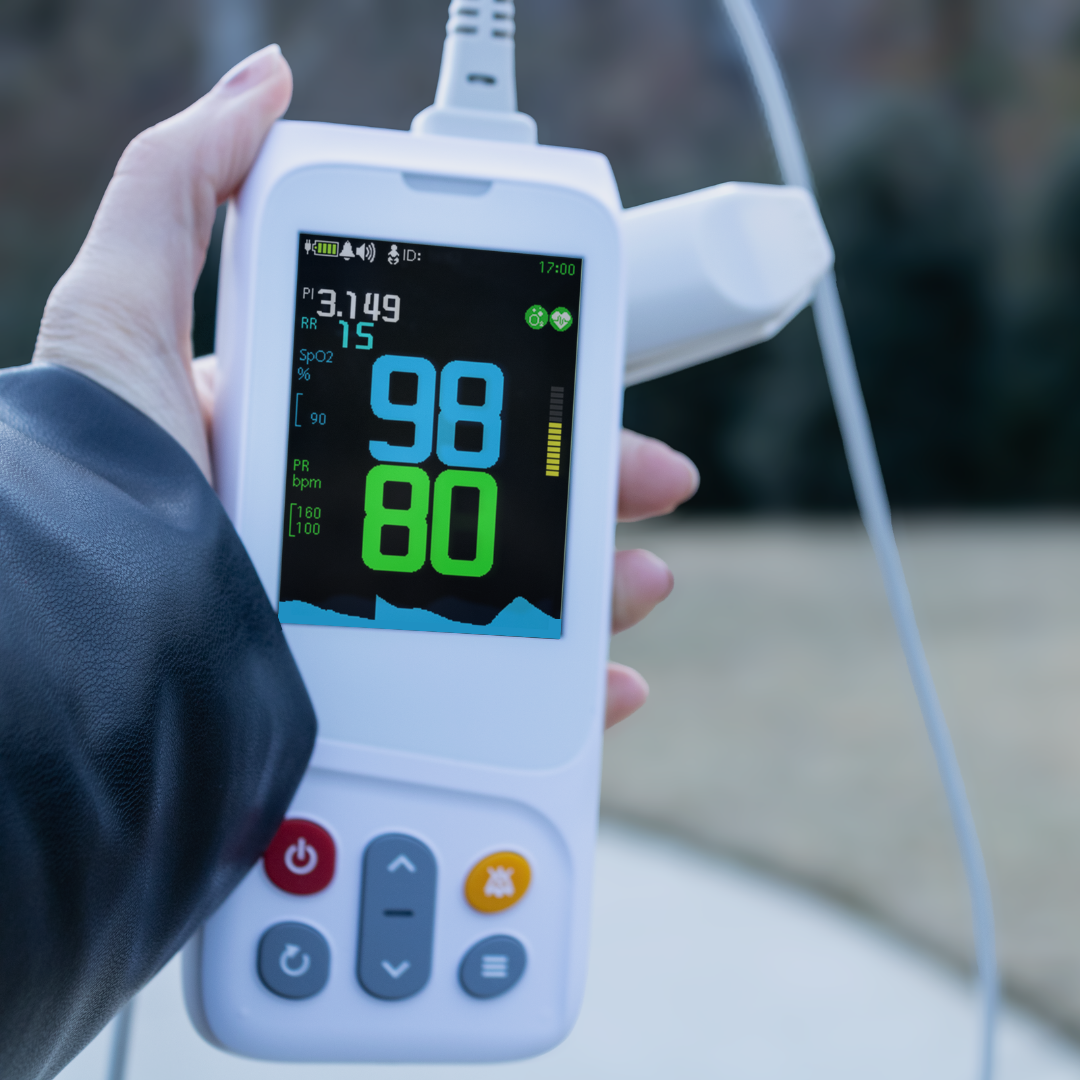
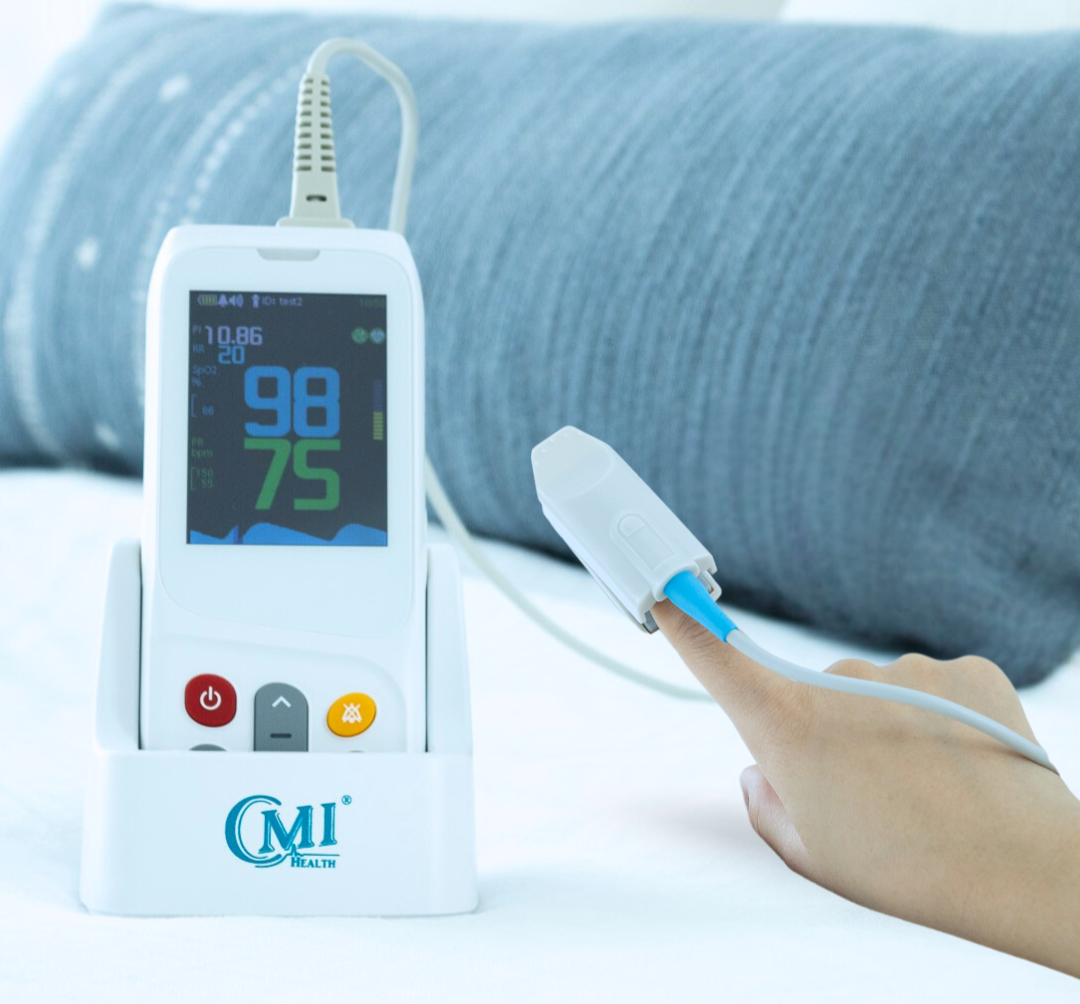
When you or a loved one’s health is involved, it is important to ensure that your medical equipment is providing accurate results. Regardless of whether you are using a Pulse Oximeter for spot-checking or continuous use, quality at-home medical equipment is key to providing you with a reliable and safe experience.
How Can I Tell if I Have a Quality Pulse Oximeter?
In the United States and internationally, there are medical standards and criteria that must be met in order for a device to achieve ISO certification (international standards) and qualify as FDA Cleared (United States Standards approved through the FDA). These standards were created to protect users and require medical device companies to manufacture and distribute equipment that is consistent, reliable, and safe. ISO certification is performed by external certifiers which are not associated with the company or manufacturer (ISO, 2021). This allows them to independently determine the certification of a product without any bias. Pulse oximeters registered as FDA-Cleared are required to meet a minimum average accuracy that is determined through desaturation studies on healthy individuals (FDA, 2021). If you purchase a pulse oximeter that does not meet these parameters, it is hard to determine how dependable it will be.
Another variable to consider is warranty protection. As a general rule of thumb, it is good to be wary of companies that do not offer warranty protection services. Warranties can ultimately help determine the quality of a product as it is a sign that the company backs their products. Click here to review CMI Health’s warranty protection policies.
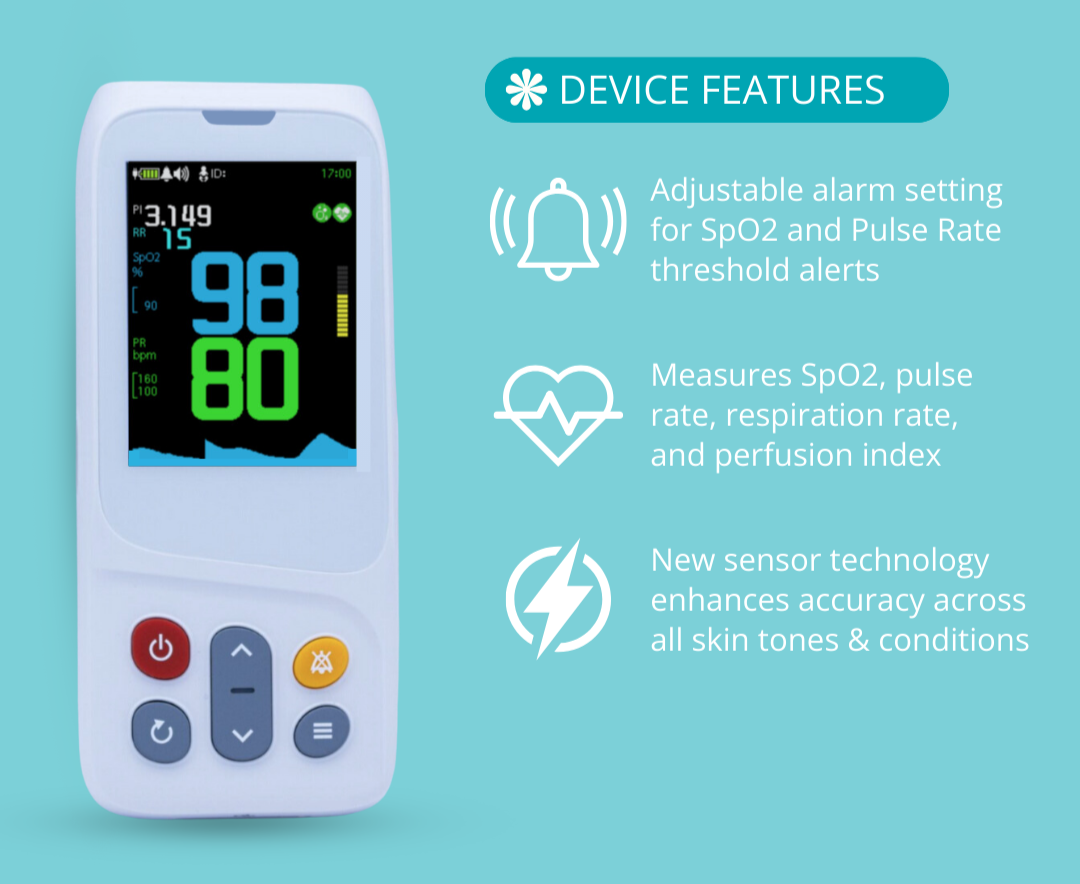
With that being said, it is important to purchase Pulse Oximeters that have been ISO certified, are FDA cleared, and have warranty protection. Quality devices should be accompanied by all of these variables, allowing the user to feel comfortable with their purchase decision. All CMI Health products are FDA Cleared, ISO certified, and covered by warranty protection.
For information on CMI Health’s ISO and FDA Compliance, click here!
What May Happen If I Purchase a Pulse Oximeter That Does Not Meet FDA or ISO Standards?
While it is hard to predict the validity of uncertified products, it is reasonable to assume that they may vary in their accuracy. In order for a pulse oximeter to be considered reliable via FDA standards, it must be able to consistently reproduce SpO2 readings within 2-3% of arterial blood gas values. Uncertified products are not required to meet this standard and therefore may produce a higher degree of variation. This leaves users at risk of not being alerted to dangerous drops in oxygen saturation levels or pulse rate.
For pulse oximeters that are not protected by warranty, users risk purchasing a faulty device with no guarantee of repair or replacement.
Interested in learning more about how accurate pulse oximeters are? Click here!























Leave a comment